The change in hCG over 48 hours gives an indication as to whether the pregnancy is likely to be ongoing (intrauterine or ectopic), or whether it is more likely to be a failing pregnancy (either intrauterine or ectopic).
#ISTAT MACHINE COST SERIAL#
The use of serial quantitative human chorionic gonadotropin (hCG) measurements is a mainstay of practice in Early Pregnancy Assessment Units (EPAUs) to aid in the management of these patients. Its incidence varies between 11 and live births in developed countries (3-5).

The main clinical concern in cases where the pregnancy location has not been determined is whether there is an ectopic pregnancy, a potentially life threatening condition. The aim of subsequent investigations is to distinguish between these outcomes. The possible clinical outcomes of a PUL are: In up to 42% of cases no intrauterine pregnancy is seen on scan (and no ectopic pregnancy is identified) and this is known as a ‘pregnancy of unknown location’ (PUL) (2). The first line of investigation for these women is an ultrasound scan to determine the location and the viability of the pregnancy (1). 50, dated July 26, 2011.In pregnant patients presenting to Primary Care or to an Early Pregnancy Assessment Unit with bleeding and pain, what is the accuracy and utility of a point-of-care quantitative hCG test compared to standard laboratory measurement?īackground, Current Practice and Advantages over Existing Technology:īleeding and pain in early pregnancy are very common presentations to Primary Care, Emergency Departments and to specialist Early Pregnancy Assessment Units. Two 9-volt lithium batteries or rechargeable battery.įactory: electronic, mechanical, thermal, pressureġ5-40☌ (59-104☏) for Medisense strip testingġ6-30☌ (61-86☏) for i-STAT cartridge testingĬomplies with U.S.11 except for deviations pursuant to laser Notice No. Test results are uploaded automatically when the i-STAT 1 handheld is placed in a downloader.Operator lockout prevents unauthorized users from performing or viewing test results.Operator and patient information can be entered via barcode scanner.The system prompts users step by step through the testing process.Patient-side testing is as easy as entering the operator and patient information into the handheld, inserting one of the several testing cartridges, and then viewing test results: features ergonomically designed soft keys for comfort and ease of use.
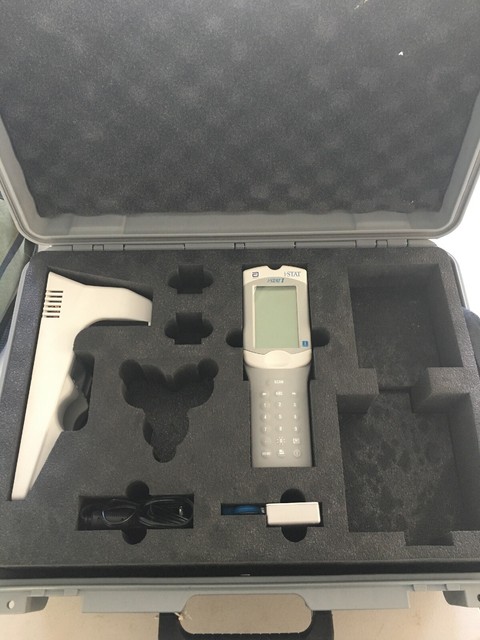
#ISTAT MACHINE COST PORTABLE#

The portable i-STAT 1 handheld makes patient-side testing easy: All other i-STAT cartridges are categorized as moderate complexity. In the USA, for the purpose of CLIA compliance, the i-STAT CHEM8+ cartridge is categorized as Waived. Analyzers, cartridges, and test strips should be used by healthcare professionals trained and certified to use the system and should be used according to the facility?s policies and procedures. The i-STAT 1 Analyzer is intended for use with i-STAT cartridges for in vitro quantification of various analytes in whole blood and with the Abbott MediSense Precision PCx Blood Glucose Test Strip for the in vitro quantification of glucose in whole blood. The Abbott i-STAT® 1 is a revolutionary handheld analyzer that provides labaccurate test results in minutes for a range of measurements including blood gases, electrolytes, chemistries, coagulation, haematology, glucose and cardiac markers. Delivers lab-quality test results to the clinician within minutes
